
You will receive two different versions: A Word document and a pdf version.įor search purposes: atoms, atomic structure, isotopes, ions, subatomic particles, lesson plan, worksheet, atomic number, atomic mass. Your download will include handouts that are ready to be copied and passed out to your students. I have used this worksheet for several purposes: a homework assignment, a quiz, classwork, and test review. This worksheet would be appropriate for any physical science or chemistry class. I use this lesson to introduce the fifth graders in my school to the basics of atomic structure. The student demonstrates an understanding of the interactions between matter and. Periodic Table Puns - Answer Key (pdf) Tips for Elements (pdf) Back to top Atoms Family (Lesson created by Kathleen Crawford, 1994) Targeted Concepts: Atoms, elements, subatomic particles, atomic structure. This worksheet is two pages long and provides multiple practice problems for your students. The resources above cover the following skills: Concepts of Physical Science (SB1, SB2, SB3, SB4) The student demonstrates an understanding of the structure and properties of matter by describing atoms and their base components (i.e., protons, neutrons, electrons). In this worksheet, students will answer questions about the following:ġ) the numbers of protons, neutrons and electrons in an atomģ) calculating the average atomic mass of an atom and its ionsĤ) the numbers of protons, neutrons and electrons in an ionġ) Students enrolled in physical science or chemistry.ģ) Students enrolled in a standard level course or an honors level course 
science cp and honors curriculum pacing guide Physical science vocabulary Mixtures work answer key. There are multiple practice problems on determining the number of protons, neutrons, and electrons in an atom as well as problems involving ions and the calculation of atomic mass. 28 Chemistry Atomic Structure Worksheet Answer Key. It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive.This review on atoms and atomic structure will provide a solid reinforcement activity for your lessons on atomic theory. 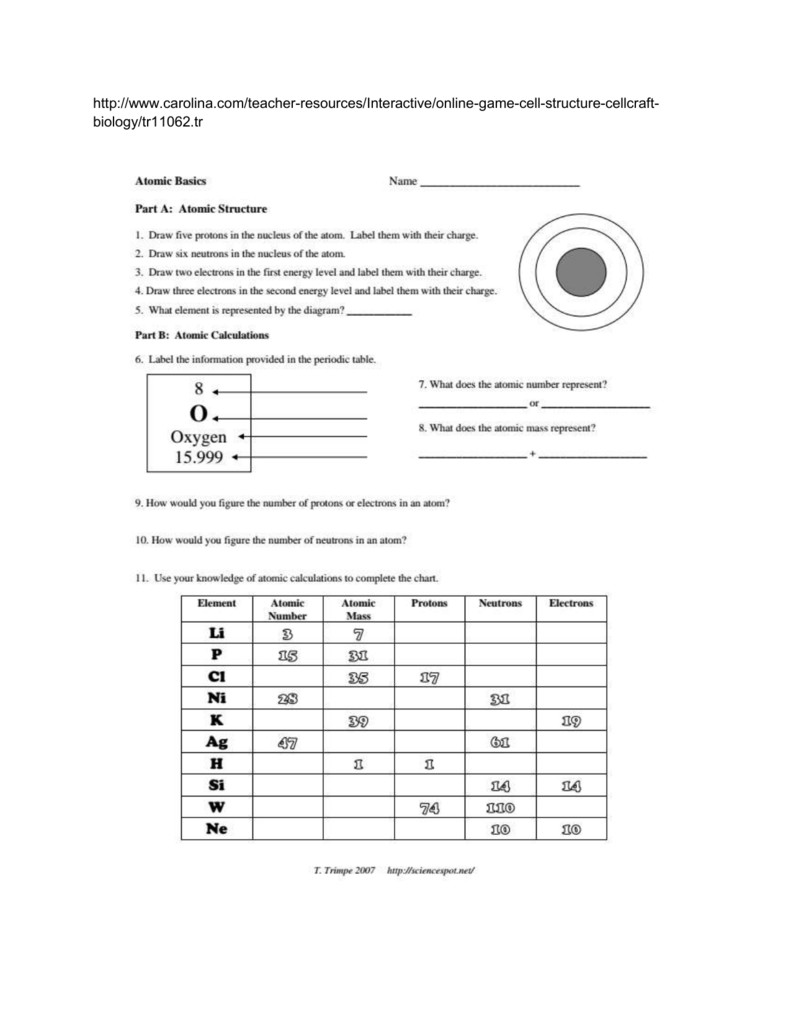
Draw three electrons in the second energy level and label them with their charge. Draw two electrons in the first energy level and label them with their charge. Draw six neutrons in the nucleus of the atom. Draw five protons in the nucleus of the atom. There are believed to be certain numbers/ratios that are inherently more stable. Atomic Basics Answer Key Part A: Atomic Structure 1. Hence, larger atoms become more unstable. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. The smallest particle into which an element can be divided and still. With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. Compared to the charge and mass of a proton, an electron has. Content covered includes describing motion, distance, Subjects: General Science, Physical Science. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. This no-prep paper AND digital paperless product is all of the notes, labs, activities, practices, and tests you need to teach your motion and force unit in your physical science class. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. The electromagnetic force of the protons wants to push them away from each other. The atomic number gives the 'identity 'of an element. It is also the number of in a neutral atom of that element. Separate answer sheets allow these resources to be used by teachers or by students during self-assessment of progress. The worksheets could be used for individual student work in class or at home. The mass of an electron is so small that it is generally not even considered. Provide a series of questions on hydrocarbons to assess learners’ knowledge and understanding of this topic at both foundation and higher levels. Here are the parts of an Atom: Electron it is a subatomic particle with a negative electrical charge. There are several forces at work inside the nuclei of atoms. Symbol Atomic Number Number of Protons Number of Neutrons Number of Electrons Mass 9 The atomic number is the number of in one atom of an element. It has a dense nucleus with a cloud of negatively charged electrons surrounding it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
